Fossey-Jouenne A., Vergne-Vaxelaire C., Zaparucha A.
J. Vis. Exp. 2018, 132
Abstract
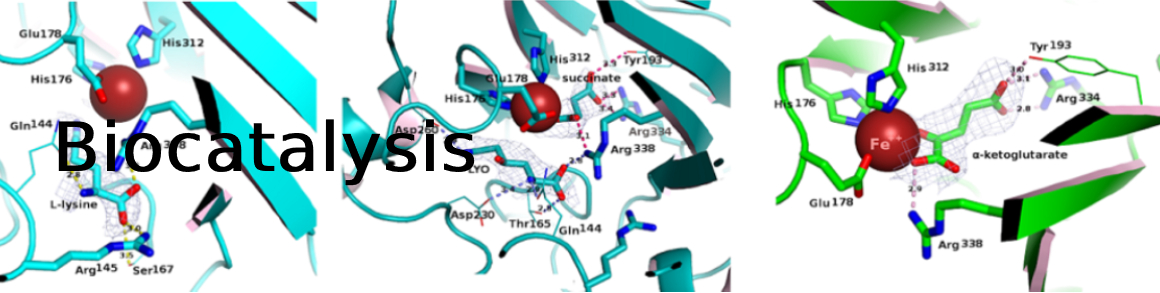
Amino alcohols are versatile compounds with a wide range of applications. For instance, they have been used as chiral scaffolds in organic synthesis. Their synthesis by conventional organic chemistry often requires tedious multi-step synthesis processes, with difficult control of the stereochemical outcome. We present a protocol to enzymatically synthetize amino alcohols starting from the readily available L-lysine in 48 h. This protocol combines two chemical reactions that are very difficult to conduct by conventional organic synthesis. In the first step, the regio- and diastereoselective oxidation of an unactivated C-H bond of the lysine side-chain is catalyzed by a dioxygenase; a second regio- and diastereoselective oxidation catalyzed by a regiodivergent dioxygenase can lead to the formation of the 1,2-diols. In the last step, the carboxylic group of the alpha amino acid is cleaved by a pyridoxal-phosphate (PLP) decarboxylase (DC). This decarboxylative step only affects the alpha carbon of the amino acid, retaining the hydroxy-substituted stereogenic center in a beta/gamma position. The resulting amino alcohols are therefore optically enriched. The protocol was successfully applied to the semipreparative-scale synthesis of four amino alcohols. Monitoring of the reactions was conducted by high performance liquid chromatography (HPLC) after derivatization by 1-fluoro-2,4-dinitrobenzene. Straightforward purification by solid-phase extraction (SPE) afforded the amino alcohols with excellent yields (93% to >95%).